|
SILO 6.4 (DRAFT)Year 6, Term 4: Cartesian geometryScope and sequence: AlgebraFocus: Coordinates |
|
Learning intention: Students explore the Cartesian coordinate system linking geometry with algebra. |
|
|
Overview: This unit starts
with a focus on René Descartes as students explore the Cartesian
coordinate system to link their prior knowledge of geometry with
new knowledge about algebra. An important algebraic concept here
is the use of letters to represent unknown values. Algebraic
equations are introduced using careful language, often avoiding
the word 'variable' unless it is warranted. For instance, if a
student is asked to find the value of 'b' when 4b = 12, b is not
a variable, but, rather, a placeholder for an unknown number
(i.e., 3).
|
|
|
NSW Syllabus
|
Australian Curriculum
|
|
"A student locates and describes
points on a coordinate plane" (MA3-GM-01).
|
"Students learn to recognise
situations, including financial contexts, that use integers;
locate and represent integers on a number line and as
coordinates on the Cartesian plane" (AC9M6N01).
|
Introduction to Cartesian geometry
René Descartes
René Descartes (1596 – 1650) could be described as a 'triple threat' as he made significant contributions to science, philosophy and mathematics. This unit is about Cartesian geometry which is a branch of mathematics also known as 'analytic geometry'. Analytic geometry is the study of geometry using a coordinate system.

(Image source https://commons.wikimedia.org/wiki/File:Frans_Hals_-_Portret_van_Ren%C3%A9_Descartes.jpg)
“It was René Descartes in 1637 who initially described the
heart as a mechanical pump and the body as a machine” (Loxley, et al.,
2018, p. 253). However, Descartes is probably best known for his
statement, “I think; therefore I am”. He
reasoned that the very act of doubting proves there must be a
'doubter' or 'thinker' (the self) for that thought or doubt to occur,
establishing the existence of the mind as an indubitable truth, even
if everything else is an illusion.

(Image source https://www.pinterest.com.au/pin/71353975316940566/)
Using coordinates
The following 10 x 10 grid is labelled with letters from left to right and numbers from top to bottom. Using a letter and number specifies particular square on the grid. For example the square in the top-left corner is A 1. Coordinates are often written in brackets and separated with a comma so that would be (A,1). The square at the bottom-right would be (J,10).

 |
Battleship |
This same grid can be used for the game Battleship. This
game is commonly available as a board game with holes and pins or as a
computer game where you play your opponent or the computer. It also
works well on paper too. You can print an A4
page here which is double sided so you can play two games. Each
player will need their own page.

Rules
- Each player secretly draws all five ships onto the grid labelled 'My fleet'. The ships must be vertical or horizontal but they cannot be diagonal or overlap.
- Decide who goes first.
- The first play calls out a pair of coordinates such as (E,4).
- If one of the other player's ships is located on (E,4) they must say, "Hit". If it is a miss then they say, "Miss". A miss is notated with an 'X' and a hit is notated with an 'O'.
- Play continues back and forth in this manner.
- When a ship has been destroyed by being hit in all squares the player must declare which ship has been sunk.
- The winner is the first player to sink all of their opponent's ships.
The photo below shows how some clips and a piece of stiff board or cardboard can enable the paper pages to fit together as a two-player board game.

This introduction to coordinates will be extended into Cartesian geometry after looking at Euclidean geometry and algebra.
Euclidean geometry
Plane geometry
The following video (3:19) is a light-hearted
look at the origins of Euclidean geometry.
The Cartesian coordinate system
The Cartesian coordinate system is a positioning system for any point on the Cartesian plane. The Cartesian plane is a plane divided into four regions (quadrants) by two perpendicular axes, the x-axis and the y-axis, which intersect at a point called the origin. The positioning system for any point in the Cartesian plane is represented by an ordered pair of real numbers (x,y), which are called the coordinates of the point relative to the origin at (0,0).
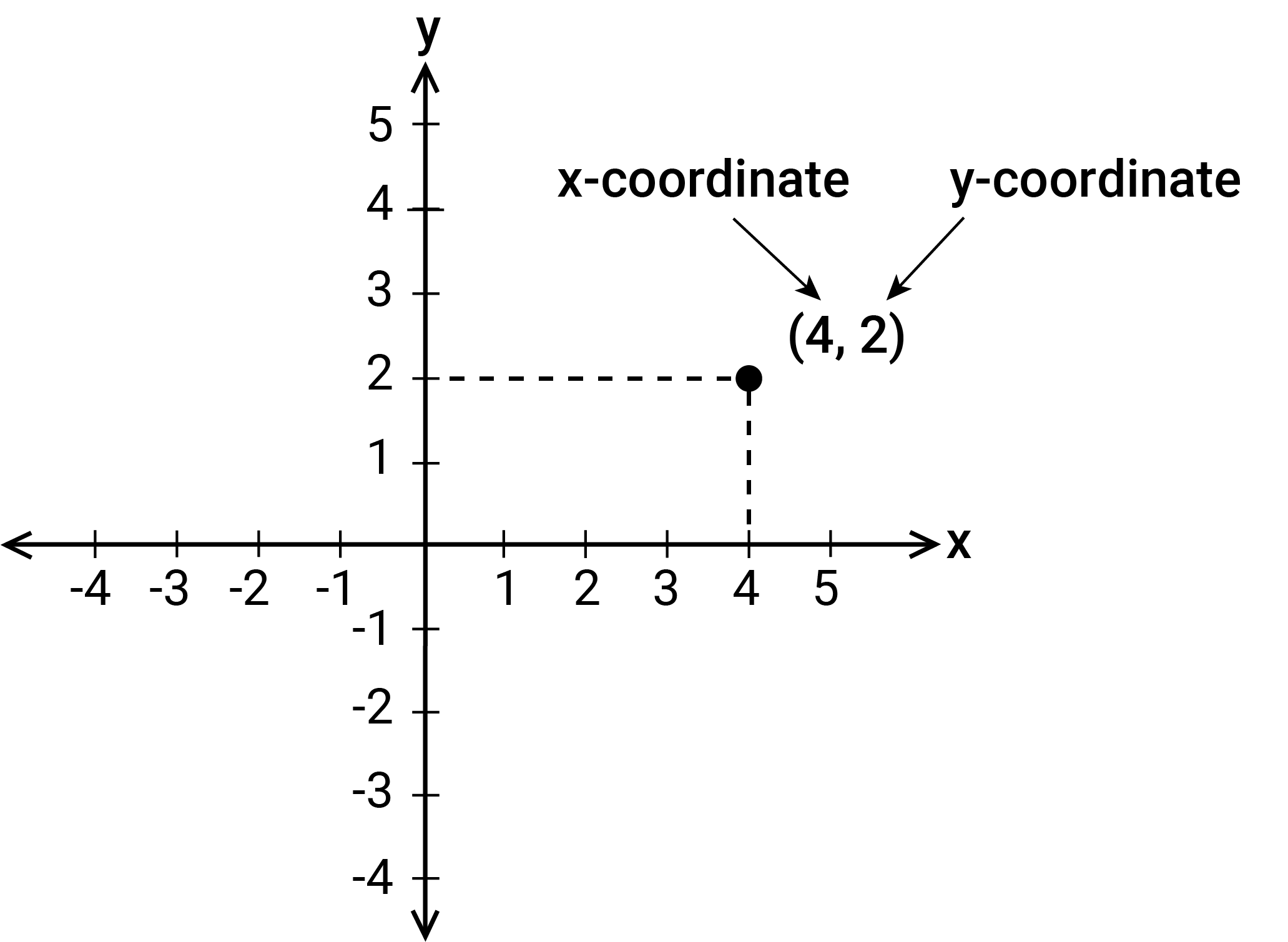
(© Australian Curriculum, Assessment and Reporting Authority (ACARA) 2010 to present, unless otherwise indicated. This image was downloaded from the Australian Curriculum website (accessed 13/02/26) and was not modified. The material is licensed under CC BY 4.0 Attribution 4.0 International Deed.)
The following image shows the Cartesian plane on a grid. Grids and graph paper are very useful when working with Cartesian coordinates.

(Image source https://socratic.org/questions/where-is-the-x-axis-and-y-axis-located)
 |
Using a table to plot coordinates |
The following image is from an A4 PDF worksheet which has table on the left and graph on the right. The double-sided worksheet can be downloaded by clicking here.
- Start by deciding on a formula. For example, y = 2x
- Calculate the y value according to the formula.
- Transfer the coordinates to the graph.

Introduction to algebra
The following video (4:49) provides an introduction to
algebra and equations using addition and subtraction.
A note about variables
Note that the video refers to x as a variable. They are using the word variable in a different way than we did in SILO 2.3 'Fair tests'. You will remember that there are many different types of variables such as independent, dependent and control. In the video they meant that x is a variable because it can change from one to question to another. However, in any given question, x simply means what is unknown but it is not really able to change. For example, in x + 3 = 9, x can only be 6 so it is not really a variable in the scientific sense.
Linking geometry with algebra
The following video (2:04) recounts a story
about how Descartes devised his coordinate system while watching a fly
on his ceiling. The story is considered to be a 'legend' as it might
not have actually occurred in this way. Other versions of the legend
mention square tiles on the ceiling which would help explain the
layout of the grid.
Algorithms and algebra
Geospacial technologies
Geospatial technologies are tools and methods for capturing, analysing, visualising, and managing location-based data. The following video (4:57) explains an exciting geospacial technology called photogrammetry.
Moderated self-assessment
Discussions with students around the key components of conceptual topics and how they fit together can generate insights into student achievement.

We welcome your feedback and suggestions
The chief investigator for The SILO Project is Associate Professor Brendan Jacobs, Head of Department STEM Education, University of New England. The SILO Project thrives on incremental improvement so constructive feedback is greatly appreciated. Please contact Brendan via email at bjacobs7@une.edu.au to share your thoughts and recommendations.

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License
Main menu

